How to accurately carry out the process of serial dilution
In serial dilution, a solute is diluted from a high concentration to a low concentration in a series of solutions. In the fields of microbiology and biochemistry, this technique is frequently employed to reduce sample concentrations to levels at which individual cells or molecules can be isolated and studied.
What is serial dilution:
In the lab, one of the most frequent methods is called “serial dilution,” and its purpose is to create a variety of dilutions from a single stock solution.
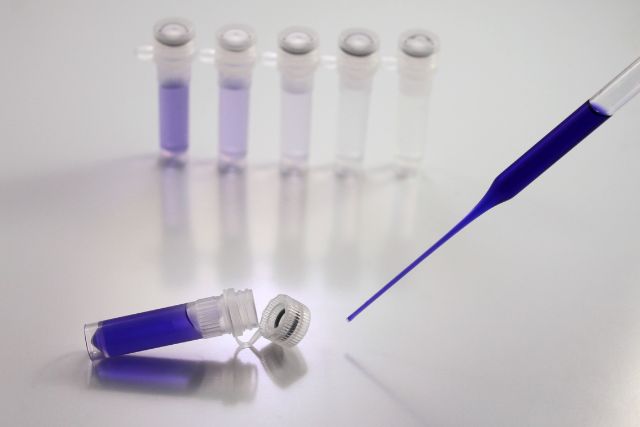
In this step of the process, you will measure out a fixed amount of the starting solution and pour it into a fresh container that already contains a fixed amount of the diluent (usually water or buffer solution). After the resultant solution has been carefully mixed to ensure that it is homogenous, a portion of this solution is taken out and transferred to the following container, which already has the same volume of diluent as the previous one. This procedure is carried out a number of times in order to produce a succession of dilutions with progressively lower concentrations.
For instance, if you have a stock solution of 10 mM and you want to prepare a series of 10-fold dilutions, you could take 1 mL of the stock solution and combine it with 9 mL of diluent to produce a solution that has a concentration of 1 mM. This would be the first step in the preparation of the dilutions. After that, take 1 millilitre of the solution that is 1 millimolar and mix it with 9 millilitres of diluent to make a solution that is 0.1 millimolar, and so on. Read More…